How to Read Probiotic Food Labels Like an Expert (2026 Guide)
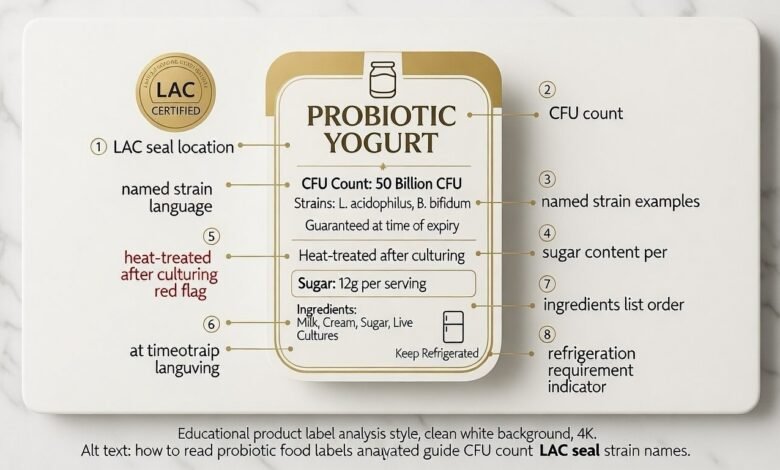
The LAC Seal — short for “Live and Active Cultures” — is an independently verified certification confirming that a dairy probiotic product contains at least 100 million CFU per gram at the time of manufacture. Unlike marketing phrases such as “contains probiotics” or “gut-friendly formula,” the LAC Seal is audited by a third party, making it one of the few trustworthy signals on a probiotic label.
The probiotic food and supplement market is worth over $70 billion globally — and a significant proportion of it is spent on products that deliver no meaningful probiotic benefit. “Contains probiotics,” “with live cultures,” “gut-friendly formula” — these phrases are marketing constructs, not regulatory standards. In most countries, the word “probiotic” can be legally applied to any product containing any bacteria, regardless of viability, count, or evidence of health benefit.
⚠ Medical Disclaimer: This article provides educational information about probiotic food label evaluation based on current research and regulatory guidance (2025–2026). It is not a substitute for medical or dietetic advice. Consult a healthcare provider before adding probiotic foods to treat a specific condition.
The result is a marketplace where genuinely effective probiotic foods sit alongside products that are probiotic in name only. Without knowing how to read probiotic food labels, the distinction is invisible. This guide gives you a precise, systematic framework for evaluating any probiotic food product in under two minutes at the shelf.
The six-point label checklist in this guide applies equally to yogurts, kefir, sauerkraut, kimchi, miso, kombucha, and probiotic supplements — giving you a universal system for confident, evidence-based probiotic shopping.
Why Probiotic Label Reading Matters
Independent laboratory testing has consistently found that a significant proportion of probiotic products fail to deliver their labelled CFU counts, with one widely cited audit finding that fewer than a third of tested products met their stated claims [2]. Knowing how to spot fake probiotic products comes down to three recurring failure patterns: heat treatment killing bacteria post-fermentation, poor cold-chain maintenance during distribution, and undisclosed use of generic bacterial strains without documented health effects. Understanding how to read probiotic food labels ensures you aren’t buying products where the bacteria were killed post-production.
The primary reasons for probiotic failure in commercial products are: heat treatment killing bacteria post-fermentation, improper cold-chain maintenance during distribution, excessive sugar content creating a competitive disadvantage for beneficial bacteria, shelf life extending beyond viable bacterial lifespan, and undisclosed use of generic bacterial strains without documented health effects.
Understanding how to read probiotic food labels does not require microbiology expertise. It requires knowing exactly which six elements to look for — and which red flags disqualify a product regardless of its marketing claims.
For the full ranked guide to probiotic foods with verified evidence, see our [10105C]- Best Probiotic Foods That Actually Work (2026 Science-Backed Guide

The 6-Point Probiotic Label Checklist
Check 1: The LAC Seal (or Equivalent Third-Party Certification)
The “Live and Active Cultures” (LAC) seal is your first green flag. It guarantees at least 100 million CFU per gram at the time of manufacture. When learning how to read probiotic food labels, look for this seal as a baseline for quality in dairy products [3].
| Certification / Seal | Jurisdiction | What It Guarantees | Products It Covers |
| LAC Seal (Live and Active Cultures) | United States | ≥100 million CFU per gram at manufacture; independent verification | Yogurt, kefir, cultured dairy |
| Soil Association Organic (with fermented claims) | United Kingdom | No added pesticides; genuine fermentation process | Organic fermented foods broadly |
| NSF International probiotic certification | International | CFU at time of expiry verified; strain identity confirmed | Supplements and functional foods |
| ISAPP-compliant labelling | International standard | Named strains, CFU at expiry, health benefit claim matches evidence | Supplements; some functional foods |
⚠ No universal regulatory seal exists for fermented vegetables (sauerkraut, kimchi) or Asian fermented foods (miso, tempeh, natto). For these products, rely on Checks 2–6 rather than seeking a certification seal.
Check 2: CFU Count — Reading It Correctly
CFU count (colony-forming units) measures viable bacteria. However, “at the time of manufacture” is a weak claim. The gold standard in how to read probiotic food labels is “CFU at time of expiry,” which accounts for natural die-off during storage [1].
| CFU Statement | What It Actually Means | Reliability | What to Look For |
| “Contains 10 billion CFU” | Count at time of manufacture; could be 1% of this by purchase date due to die-off | LOW | Avoid without an expiry date reference |
| “10 billion CFU at time of expiry” | Count guaranteed until best-before date; accounts for storage die-off | HIGH — preferred | This is the gold standard statement |
| “10 billion CFU per serving” | Serving size must be checked — some products set a “serving” at 2–3 times normal consumption | MODERATE | Verify serving size matches realistic consumption |
| “Contains probiotics” with no CFU | No quantifiable information; regulatory minimum not met for any evidence-based claim | VERY LOW | Disqualify the product |
💡 Minimum thresholds by use: For general gut health maintenance, 1 billion CFU per serving is adequate for most adults. For post-antibiotic recovery, 10–50 billion CFU is recommended. For specific conditions (IBS, C. difficile prevention) — 50+ billion CFU under medical guidance. More is not always better; match dose to purpose.
CFU at Expiry vs Manufacture — And How to Spot the Difference at a Glance The single most common probiotic label red flag that shoppers miss is the phrase “at time of manufacture” — which legally permits a product to state 50 billion CFU even if fewer than 1 billion survive to the shelf. How to read probiotic labels for this specific detail: scan the CFU statement for the words “through expiry,” “at end of shelf life,” or “guaranteed until best before date.” If none of these phrases appear, the CFU figure is a manufacturing number, not a consumption number. Named probiotic strains vs generic live cultures follow the same logic: a product listing “Lactobacillus rhamnosus GG” is making a verifiable, strain-specific claim; one listing “Lactobacillus cultures” is not — and cannot support any specific health benefit claim regardless of the CFU stated.
For more information, explore our article [Best Probiotic Foods That Actually Work]
Check 3: Named Strains — The Most Important Check
A genus name like Lactobacillus isn’t enough. Experts in how to read probiotic food labels look for the full three-part name (e.g., Lactobacillus rhamnosus GG). Only named strains have verifiable clinical evidence [4]
| Naming Level | Example | What You Know | Health Claim Validity |
| Genus only | Lactobacillus | Almost nothing — thousands of species and millions of strains exist | Cannot be verified |
| Genus + Species | Lactobacillus acidophilus | Species-level properties — still broad; thousands of strains per species | Very limited |
| Full strain name | Lactobacillus acidophilus NCFM | Specific strain with its own research record, documented mechanisms, and clinical trials | Verifiable — strong basis for claims |
| Trademarked strain name | LGG (= L. rhamnosus GG), BB-12 (= B. animalis lactis) | Specific, extensively studied strains with decades of clinical evidence | Highly verifiable — best evidence-based |
The best probiotic strains to look for on labels — those with the strongest published research records in commercial foods and supplements — are: Lactobacillus rhamnosus GG (LGG), Bifidobacterium animalis lactis BB-12, Lactobacillus acidophilus NCFM, Bifidobacterium longum BB536, and Saccharomyces boulardii CNCM I-745. Any product listing one of these by its full trademarked name can be directly assessed against decades of clinical trial evidence.
Check 4: Ingredients Order and Red-Flag Additives
What does “heat-treated after culturing” mean on a probiotic label? It means the manufacturer cultured live bacteria during production — then deliberately applied heat to kill them before packaging, typically to extend shelf life or stabilise texture. The result is a product that was technically fermented but contains zero live bacteria at the point of consumption. This is a total disqualifier for any probiotic purpose, regardless of all other label claims. Also, avoid emulsifiers like Polysorbate 80, which can damage the gut lining. High-quality probiotic foods usually have five or fewer recognizable ingredients [5]
| Ingredient / Feature | What It Signals | Action |
| “Heat-treated after culturing” on label | All bacteria are deliberately killed post-production; zero live probiotic benefit | Disqualify immediately — do not purchase for probiotic purposes |
| Polysorbate 80 (E433) or carboxymethylcellulose (E466) in ingredients | Emulsifiers that dissolve gut mucus — directly counteract the gut health benefits of probiotics | Avoiding these additives undermines the purpose of taking probiotics |
| Sorbitol (E420), mannitol (E421), and sucralose in significant amounts | Artificial sweeteners disrupt the microbiome, may cause diarrhoea, and counteract probiotic colonisation | Avoid in probiotic products specifically |
| Added sugar above 8g per serving | Feeds pathogenic bacteria competitively; reduces net probiotic benefit | Downgrade assessment; acceptable only in small doses |
| Carrageenan (E407) | Promotes intestinal inflammation; directly damages the gut barrier. Probiotics are meant to support | Disqualify for gut health purposes |
| Short ingredients list (5 or fewer recognisable ingredients) | Indicates minimal processing; maximises likelihood of genuine probiotic viability | Positive signal — prefer |

Check 5: Storage and Packaging Requirements
The cold chain is essential for bacterial viability. Most probiotic bacteria — including all Lactobacillus and Bifidobacterium species used in probiotic foods — require refrigeration to maintain viability. At room temperature, bacterial die-off in dairy products occurs at rates of 10–20% per week; shelf-stable products claiming live cultures require either very high starting counts, protective packaging, or spore-forming strains.
| Storage Type | Viable Probiotic Strains | Products | Buyer’s Assessment |
| Refrigerated (0–4°C / 32–39°F) | Lactobacillus, Bifidobacterium, Leuconostoc, most food-based strains | Kefir, yogurt, raw sauerkraut, kimchi, fresh miso, tempeh | Highest confidence in viability — preferred |
| Ambient shelf-stable (room temperature) | Spore-forming strains only (Bacillus subtilis, B. coagulans); specialist protective coatings | Natto (B. subtilis naturally spore-forming), some supplements | Verify strain is genuinely heat-resistant; most generic “probiotic” drinks fail here |
| Frozen | Most strains survive freezing well if frozen quickly post-culture | Frozen kefir, some yogurt | Acceptable — check that freezing occurred post-culturing, not post-pasteurisation |
⚠ Shelf-stable “probiotic” drinks in ambient packaging (e.g., many flavoured probiotic shots or shelf-stable yogurt alternatives) rarely contain viable bacteria of meaningful species. The LAC seal is not awarded to shelf-stable products. Treat shelf-stable probiotic food claims with significant scepticism unless the strain is explicitly a spore-former.
💡 For Complete information, explore the complete segments of our Nutrition and Gut-Brain Health Series Overview
Check 6: Best-Before Date and Batch Traceability
Fresh is best for all probiotic foods. Bacterial counts in dairy ferments decline approximately 10–30% per week after production at optimal refrigeration. Buying the product with the longest remaining shelf life maximises the CFU count you actually consume. For fermented vegetables, recent production dates indicate more active fermentation, though established sauerkraut and kimchi maintain live bacteria well in brine for 3–6 months under refrigeration.
- Always check “the “best-before date — choose the latest date available
- Batch traceability (lot numbers) indicates quality-controlled production — a positive signal for supplements specifically
- For homemade ferments: sauerkraut and kimchi reach peak probiotic activity at 1–4 weeks of fermentation at room temperature; refrigerate when the desired flavour is reached and consume within 3 months
Product Category Quick-Reference Guide
| Category | Key Checks | Instant Disqualifiers | Best Signals |
| Yogurt | LAC seal; named strains; sugar < 8g; full-fat preferred | Heat-treated after culturing; emulsifiers in ingredients | LAC seal + named strains + plain or lightly flavoured |
| Kefir | Refrigerated; slight carbonation; plain unsweetened | Shelf-stable; UHT-treated; fruit-flavoured with >12g sugar | “Kefir grains fermented” language; LAC seal; 30+ strains claimed |
| Sauerkraut / Kimchi | Unpasteurised label; refrigerated; simple ingredients (vegetables + salt) | Canned; shelf-stable; contains vinegar (indicates no fermentation) | “Raw” or “unpasteurised” label; brine visible; refrigerated section |
| Miso | Refrigerated; unpasteurised; rice/barley/soy + salt only | Instant miso packets; long shelf-stable shelf life | Refrigerated; traditional Japanese production; 6+ months fermentation stated |
| Kombucha | Active cultures stated; refrigerated; <5g sugar per 240ml | Pasteurised; shelf-stable; >15g sugar per bottle | “Raw” or “live” label; visible sediment; light effervescence |
| Probiotic supplements | CFU at expiry stated; named strains; third-party tested | CFU at manufacture only; no strain names; ambient storage | NSF or ISAPP-compliant; named trademarked strains; enteric coating where needed |
5-Second Shelf Test: Making the Decision Quickly
When you have 5 seconds at the supermarket shelf — not 5 minutes — use this rapid elimination sequence:
- Is it refrigerated? — If not, disqualify unless it is natto or a verified spore-former supplement
- Is there a LAC seal or equivalent third-party mark? — If yes, proceed with confidence for dairy products
- Does the label mention any strain by full name (genus + species + strain code)? — If yes, it is a genuine probiotic claim
- Is “heat-treated after culturing” present? — If yes, put it back immediately
- Is added sugar under 8g per serving? — If over, factor it as a partial disqualifier
💡 The single fastest heuristic for probiotic food quality: if the only information about bacteria is “contains live cultures” or “probiotic formula” with no named strains, no CFU count, and no seal — the product is probiotic in marketing only.
For the complete framework of how to integrate verified probiotic foods into daily gut-healthy eating, see our pillar guide: Gut Health Diet: Essential Foods and Tips

Key Takeaways
- “Probiotic” is unregulated in most countries — a product can carry this label regardless of bacterial viability, count, or health evidence. Label-reading skills are essential for value and efficacy.
- The six-point checklist (LAC seal, CFU at expiry, named strains, ingredients check, refrigerated storage, and fresh best-before date) provides a complete evaluation framework applicable to all probiotic food categories.
- “Heat-treated after culturing” is the single most important red flag — it means all bacteria were deliberately killed post-fermentation, delivering zero probiotic benefit regardless of all other label claims.
- CFU at time of expiry is the meaningful guarantee; CFU at time of manufacture is a marketing figure that overstates the count you will actually consume.
- Named strains (genus + species + strain code, or trademarked strain names) are the minimum standard for verifiable health claims; generic “live cultures” labelling does not support specific health benefit claims.
- The 5-second shelf test — refrigerated → LAC seal → named strain → no heat-treatment → sugar under 8g — provides a rapid-decision framework for confident probiotic shopping.
Final Thoughts
You now have a systematic, evidence-based framework for evaluating any probiotic food product in under two minutes. The probiotic food market rewards informed buyers — the products that deliver genuine benefit exist alongside those that do not, often on the same shelf at similar prices.
Next time you are in the supermarket, apply the 5-second shelf test to the yogurt or kefir in your basket. If it passes all five checks, you have a genuine probiotic food. If it fails on “heat-treated after culturing” or has no named strains, put it back and look for one that meets both criteria.
Save this guide as a reference for your next shop. Share it with family and friends who are spending money on probiotic products — helping them become informed buyers is one of the most practical gut health gifts you can give.
FAQ
Q1: Is a higher CFU count always better?
A: Not necessarily. A lower dose of a named strain with clinical evidence is better than a high dose of an anonymous strain.
Q2: What does “at time of manufacture” vs “at time of expiry” mean on labels?
A: At the time of manufacture,” means the CFU was measured when the product was made — bacteria die during storage, transport, and shelf time, so the count at the moment of consumption could be a small fraction of what is stated. “At time of expiry” (or “at end of shelf life”) means the stated CFU count is guaranteed to still be present on the best-before date, accounting for storage die-off during the guaranteed shelf period. Always prefer products with the “at time of expiry” guarantee.
Q3: Do probiotic drinks like Yakult work?
A: Yes, Yakult is a genuine probiotic containing the well-studied L. casei Shirota strain (6.5bn CFU), which supports immunity and transit. However, its 11g of sugar per 65ml bottle can feed pathogens, potentially counteracting the benefits. Lower-sugar alternatives like plain kefir offer better value for gut health [4]
Q4: Why do some labels only say “live cultures”?
A: Live cultures” means the bacteria are alive, but no specific health benefit is claimed. “Probiotic” implies a defined health benefit, which requires more evidence.
Q5: Can I tell if a probiotic food is still active without laboratory testing?
A: Kefir & Kombucha: Look for slight carbonation, bubbles, or effervescence upon opening; “flat” liquids suggest inactive cultures.
Sauerkraut: Agitating the jar should produce tiny bubbles in the brine; clear, “lifeless” brine indicates lost activity.
Yogurt: Should have a clean, tangy aroma and slight resistance when stirred.
Supplements (Mix one capsule into 240ml of warm milk (37°C) and leave for 24 hours.) and leave for 24 hours. Active Lactobacillus will curdle the milk; no change means the bacteria are non-viable.
Nutrition and Gut-Brain Health
This article is part of the Comprehensive Gut Health & Nutrition Series — an evidence-based collection of guides exploring the gut microbiome, digestive health strategies, and the direct connection between nutrition and mental and physical performance.
→ View all Nutrition and Gut-Brain Health series articles here
External Resources
- [1] ISAPP: What Is a Probiotic?
https://isappscience.org/for-consumers/learn/probiotics/ - [2] NIH/PMC: Probiotic Label Accuracy and CFU Verification (2023): https://pmc.ncbi.nlm.nih.gov/articles/PMC10443604/
- [3] FDA: Guidance for Industry — Labeling of Dietary Supplements Containing Live Microbials: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/guidance-industry-labeling-dietary-supplements-containing-live-microbials.
- [4] British Dietetic Association (BDA): Probiotics and Gut Health:
https://www.bda.uk.com/resource/probiotics.html - [5] Dietitians of Canada: Eating Well with Fermented Foods: https://www.dietitians.ca/Downloads/Factsheets/Fermented-Foods-and-Your-Gut.aspx.





